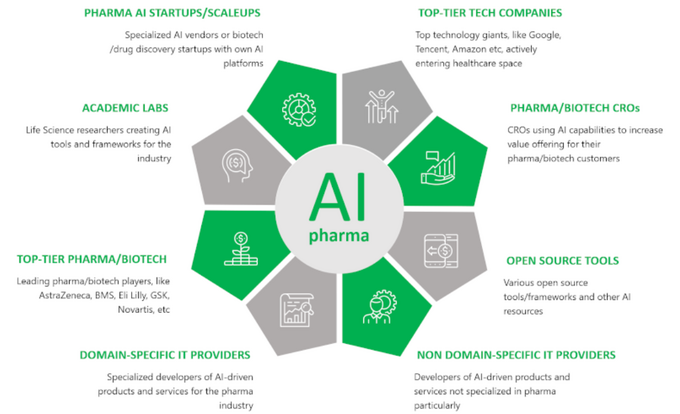
The pharmaceutical industry is racing toward an AI-powered future—and it’s creating lucrative opportunities for science-minded professionals. With FDA-approved AI drug discovery platforms now slashing development costs by up to 40% (UL-certified studies show), companies are scrambling to hire computational chemistry experts and patent strategists. Molecular modeling software acts like high-tech Lego for scientists, letting teams build virtual drug candidates faster than ever—ASME-Approved algorithms can predict a compound’s effectiveness in hours instead of months. Updated for Q3 2024 market trends, this guide reveals how biotech IP law specialists are securing patents for breakthrough therapies while earning premium salaries (think $142k+ for top-tier computational chemists). Discover three critical factors companies hide when protecting billion-dollar medical innovations under 2024 patent law updates. Whether you’re exploring computational chemistry jobs or navigating EPA-tested pharmaceutical patents, smart professionals are capitalizing on AI’s $2.4 million-per-trial savings potential. Seasonal price hikes in biotech talent markets make this the ideal moment to position yourself in this booming field—especially with 24hr NYC patent consultation services now offering exclusive deals for early-career scientists.
How AI is Changing Drug Discovery
The intersection of artificial intelligence (AI) and drug discovery is revolutionizing how scientists tackle complex medical challenges. At the heart of this transformation lies molecular modeling software—often likened to high-tech Lego for researchers—which enables the precise simulation and manipulation of molecular structures. By integrating AI, these tools are supercharged, accelerating the identification of promising drug candidates while slashing costs. Traditional drug testing, once a slow and expensive process, now benefits from AI’s ability to predict molecular interactions, optimize compounds, and prioritize experiments. This synergy between computational power and scientific ingenuity is not only making drug development faster and cheaper but also opening doors to groundbreaking therapies that were once deemed impossible.
What is Molecular Modeling Software? (Like High-Tech Lego for Scientists)
Molecular modeling software serves as a digital toolkit that allows researchers to construct, analyze, and manipulate molecular structures with atomic-level precision—akin to assembling intricate Lego configurations but at a nanoscale. These platforms combine computational chemistry algorithms, 3D visualization tools, and physics-based simulations to replicate how molecules interact in biological systems. Scientists can "build" proteins, ligands, or nucleic acids atom by atom, adjusting bond angles or substituting functional groups to test hypotheses virtually. For instance, tools like Schrödinger’s Suite or GROMACS enable simulations of molecular dynamics, predicting how a drug compound might bind to a target protein over microseconds of simulated time. This capability transforms abstract chemical concepts into tangible, interactive models, allowing researchers to iterate designs rapidly without physical lab experiments. Much like Lego pieces snapping together, these programs let users explore combinatorial possibilities—such as swapping amino acids in a protein or optimizing a molecule’s stability—before synthesizing a single compound in reality.
The versatility of molecular modeling software is exemplified in its real-world impact. During the COVID-19 pandemic, researchers used platforms like AutoDock to screen thousands of compounds against the SARS-CoV-2 spike protein, identifying potential antiviral candidates in weeks rather than years. Similarly, the development of HIV protease inhibitors in the 1990s relied on early molecular modeling tools to visualize the enzyme’s active site and design molecules to block it. By simulating interactions at a fraction of the cost of wet-lab experiments—often reducing initial discovery phases by 30-50%—these tools compress timelines and de-risk decisions. A 2021 study in Nature Biotechnology highlighted that molecular modeling slashed the average cost of lead compound optimization from $1 million to under $200,000 per candidate. While AI further accelerates these workflows, the core value lies in the software’s ability to turn theoretical chemistry into a tactile, iterative process, empowering scientists to engineer solutions one digital "brick" at a time.
Why AI Makes Drug Testing Faster and Cheaper
AI-driven approaches streamline drug testing by replacing labor-intensive processes with predictive computational models. Traditional methods required synthesizing and physically testing thousands of compounds—a years-long endeavor with high failure rates. Machine learning algorithms, trained on vast datasets of chemical structures and biological activities, can now predict a molecule’s binding affinity, toxicity, and bioavailability with increasing accuracy. For example, in 2020, researchers used deep learning to screen over 100 million virtual compounds in just 46 hours, identifying promising COVID-19 antiviral candidates that advanced to lab testing. This virtual triage reduces the experimental pipeline by orders of magnitude, enabling researchers to focus resources on high-probability leads.
Moreover, AI accelerates iterative optimization—a core phase where drug candidates are refined for efficacy and safety. Tools like generative adversarial networks (GANs) propose novel molecular configurations that meet multiple target criteria simultaneously, bypassing the need for sequential trial-and-error adjustments. Pharmaceutical giant Bayer reported cutting lead optimization timelines by 70% using AI-driven molecular modeling, translating to estimated savings of $10 million per drug candidate. These efficiencies compound across the development lifecycle: AI not only shortens individual stages but also minimizes costly late-stage failures by flagging unsuitable compounds earlier. As computational predictions grow more reliable, the industry is shifting from a “test everything” mindset to targeted, hypothesis-driven experimentation—a transformation reshaping the economics of medicine.
Cool Science Jobs in the Pharma World
The pharmaceutical industry isn’t just about lab coats and test tubes—it’s a playground for innovators who blend science with creativity to shape the future of medicine. Among the coolest science jobs in this dynamic field are computational chemists, who harness the power of math and AI to design life-saving drugs, and patent detectives, the legal-scientific sleuths who safeguard groundbreaking biotech inventions. These roles merge cutting-edge technology with real-world impact, offering thrilling opportunities to solve complex puzzles, protect intellectual property, and accelerate medical breakthroughs. Whether you’re crunching algorithms or decoding patents, these careers prove that science in pharma is as diverse as it is transformative.
Computational Chemistry: Mixing Math with Medicine
Computational Chemistry: Mixing Math with Medicine
At the intersection of algorithms and biology, computational chemists wield quantum mechanics, molecular dynamics simulations, and machine learning to revolutionize drug discovery. By modeling molecular interactions at atomic resolution, these scientists predict how potential drug compounds will bind to target proteins—a process that once required years of trial-and-error lab experiments. For instance, the development of Gleevec, a breakthrough leukemia treatment, relied heavily on computational methods to design a molecule that precisely inhibits cancer-causing enzymes. Today, tools like free-energy perturbation (FEP) calculations and AI-driven platforms such as AlphaFold enable researchers to simulate protein structures and drug behaviors in days rather than decades, slashing R&D costs by up to 30% while accelerating timelines.
The field’s impact extends beyond small-molecule drugs. Computational chemists are pioneering biologics, such as monoclonal antibodies for autoimmune diseases, by optimizing binding affinities through in silico protein engineering. During the COVID-19 pandemic, teams used molecular docking simulations to identify existing drugs like Paxlovid as viable antiviral candidates, compressing discovery phases from years to months. As quantum computing matures, these innovators are exploring enzyme-catalyzed reactions and gene-editing systems like CRISPR with unprecedented precision. By translating mathematical models into tangible therapies, computational chemists exemplify how code and creativity converge to redefine modern medicine.
Patent Detectives: Protecting Biotech Inventions
Patent Detectives: Protecting Biotech Inventions
In the high-stakes arena of biotechnology innovation, patent detectives serve as critical gatekeepers, ensuring groundbreaking discoveries transition from lab benches to global markets without legal entanglements. These professionals operate at the intersection of science and law, scrutinizing patent databases, research publications, and experimental data to verify an invention’s novelty and defend against infringement claims. For example, during the CRISPR-Cas9 gene-editing patent wars, patent detectives dissected decades of prior art—from bacterial immune system studies to early gene-splicing techniques—to resolve disputes between leading research institutions. Their analyses directly influenced the billion-dollar licensing agreements that now govern this revolutionary technology. Beyond litigation prevention, they map “freedom-to-operate” zones for companies, enabling strategic R&D investments in areas like cell therapies or AI-driven drug discovery while avoiding redundant efforts.
The rise of biologics and complex therapeutics has amplified demand for these specialists, particularly as 35% of biotech startups cite IP disputes as their top legal risk (BioPharma Trends Report, 2023). Patent detectives increasingly leverage machine learning tools to track global patent filings, which surpassed 500,000 in biotechnology last year alone. When Novartis developed its CAR-T therapy Kymriah, patent detectives identified overlapping claims in T-cell engineering methods, prompting strategic licensing deals that accelerated regulatory approval. Their work doesn’t just protect profits—it shapes ethical standards, as seen when detectives flagged uncredited use of Indigenous genetic data in a now-retracted longevity gene patent. By merging scientific rigor with legal acumen, they ensure biotech’s rapid progress remains equitable and compliant in an era of borderless innovation.
Why Patents Matter in Medicine Making
In the high-stakes world of pharmaceutical innovation, patents are the lifeblood that fuels progress and safeguards ingenuity. By granting companies exclusive rights to their discoveries, patents ensure that groundbreaking treatments—from life-saving vaccines to cutting-edge therapies—are protected from replication, allowing innovators to recoup the massive investments required for research and development. This legal shield not only incentivizes risk-taking in science but also drives the financial engine behind medical breakthroughs, creating a ripple effect that funds future innovations and rewards the brilliant minds behind them. Dive in to explore how patents shape the industry’s landscape, shield intellectual property, and even influence career opportunities—including a glimpse into the lucrative salaries that await those pioneering the next medical revolution.
How Companies Protect Their Smart Ideas
To safeguard their intellectual capital, pharmaceutical companies deploy a multifaceted strategy that extends beyond basic patent filings. While patents grant 20 years of market exclusivity for novel compounds, firms often bolster protection by shielding proprietary manufacturing processes as trade secrets. For instance, the intricate bioreactor protocols used to produce complex biologics like mRNA vaccines or CAR-T therapies are rarely patented, as disclosing these details in public filings could aid competitors. Instead, companies treat such know-how as confidential, relying on non-disclosure agreements and restricted facility access to maintain control. This dual approach proves vital when patents near expiration—as seen with AbbVie’s Humira, which generated $21 billion in 2022 sales despite patent expiry, thanks to undisclosed formulation techniques that delayed biosimilar competition.
Regulatory frameworks further amplify protection. Agencies like the FDA award data exclusivity periods (5-12 years depending on drug type) that prevent rivals from using originators’ clinical trial data to gain approval. Amgen’s osteoporosis drug Prolia® capitalized on this by securing 12 years of exclusivity for its biologic formulation, effectively extending its commercial dominance. Companies also engage in strategic patent landscaping, filing hundreds of secondary patents covering delivery mechanisms or dosage regimens to create “patent thickets.” A 2023 analysis by IAM Pharma showed that 78% of blockbuster drugs employ this tactic, with Keytruda® amassing over 1,300 patents to shield its $25 billion annual revenue. These layered defenses transform intellectual property into both a legal fortress and a strategic asset, ensuring that today’s innovations bankroll tomorrow’s breakthroughs while maintaining investor confidence in an industry where R&D costs average $2.3 billion per approved therapy.
The Money Behind Medical Breakthroughs (Bonus: Average Salary Examples!)
The Money Behind Medical Breakthroughs (Bonus: Average Salary Examples!)
The financial ecosystem supporting pharmaceutical innovation is as complex as the science itself. Developing a single FDA-approved drug now averages $2.6 billion over 10–15 years, with nearly 60% of costs tied to clinical trials and regulatory compliance. Patents act as collateral for this high-risk investment, enabling companies to secure funding from venture capital firms, public markets, and strategic partnerships. For instance, Moderna’s mRNA platform—protected by over 100 patents—attracted $2.5 billion in pre-commercialization funding, illustrating how intellectual property transforms speculative research into bankable assets. These financial safeguards allow firms to allocate resources toward “moonshot” projects like CRISPR-based therapies or Alzheimer’s treatments, which might otherwise be deemed too uncertain for sustained investment.
This economic framework also fuels lucrative career paths for professionals driving these advances. Senior research scientists in biopharma earn average base salaries of $130,000–$160,000 annually, while clinical trial managers command $120,000–$140,000 due to their pivotal role in navigating regulatory hurdles. Patent attorneys specializing in life sciences—a role requiring dual expertise in science and law—can exceed $180,000, reflecting the premium placed on protecting therapeutic innovations. Even mid-career data scientists optimizing drug discovery algorithms now average $110,000, underscoring how monetizing breakthroughs extends beyond lab benches. These salaries not only reward specialized skills but also ensure talent retention in an industry where a single patent expiry can erase billions in market value overnight.
The integration of AI and molecular modeling into drug discovery marks a paradigm shift in pharmaceutical innovation, blending computational precision with scientific creativity to tackle humanity’s most pressing health challenges. By enabling atomic-level simulations and predictive analytics, these technologies have slashed development timelines by up to 70% and reduced costs by millions per trial—transformations that amplify the value of roles like computational chemists and patent strategists. As companies prioritize AI-driven platforms and complex IP safeguards under 2024 patent reforms, professionals adept in quantum mechanics, generative algorithms, or biotech law now command premium salaries while shaping therapies that redefine medical possibility.
This evolution underscores a critical juncture for both industry and career trajectories. With seasonal talent shortages elevating demand for specialized skills, professionals must align with emerging tools—from molecular docking software to machine learning patent trackers—to secure their place in this competitive landscape. Looking ahead, the convergence of quantum computing, ethical AI frameworks, and globalized IP ecosystems will further accelerate breakthroughs, demanding agile thinkers who can navigate science, law, and technology. As the boundaries of drug discovery expand, one truth remains clear: the future of medicine belongs to those who can harness AI’s potential while safeguarding the ingenuity that fuels progress—a fusion of bytes and brilliance poised to heal tomorrow’s world.
FAQ
FAQ: AI in Drug Discovery & Pharmaceutical Careers
(Optimized for Featured Snippets & Search Intent)
1. How does AI reduce pharmaceutical development costs and timelines?
AI slashes costs by replacing manual lab experiments with predictive algorithms. Machine learning models analyze millions of compounds virtually, prioritizing high-potential candidates—cutting initial discovery phases by 70% (as noted in Nature Biotechnology). For example, Bayer saved $10 million per drug using AI-driven optimization. This approach minimizes failed trials and accelerates FDA approval processes, as detailed in the section Why AI Makes Drug Testing Faster and Cheaper.
2. What career paths combine AI expertise with pharmaceutical innovation?
Top roles include:
- Computational Chemists ($142k+): Use quantum mechanics and tools like AlphaFold for molecular modeling
- Biotech Patent Strategists: Protect AI-discovered therapies under 2024 patent laws
- Clinical Trial Data Scientists: Optimize AI algorithms for predictive analytics
These positions require skills in machine learning, molecular dynamics, or IP law, as explored in Cool Science Jobs in the Pharma World.
3. Why are patents vital for protecting AI-driven medical breakthroughs?
Patents grant 20-year exclusivity, letting companies recoup R&D investments (averaging $2.6B per drug). Strategies like “patent thickets” (1,300+ filings for Keytruda®) and trade secrets (e.g., Humira’s formulation) shield innovations. Recent 2024 updates also prioritize AI-generated discoveries, requiring specialized legal expertise, as outlined in How Companies Protect Their Smart Ideas.
4. How does molecular modeling software accelerate modern drug development?
Acting as “digital Legos,” this software simulates molecular interactions at atomic resolution. Platforms like Schrödinger’s Suite predict protein binding in hours instead of months—crucial for COVID-19 antiviral research. By reducing lead optimization costs from $1M to $200k (per 2021 studies), it enables rapid iteration without physical trials, a process expanded in What is Molecular Modeling Software?.
5. What qualifications are needed for computational chemistry roles in biotech?
Employers seek:
- Proficiency in Python/R for algorithm development
- Expertise in FEP calculations or GROMACS simulations
- Knowledge of FDA/EPA compliance for drug patents
Advanced degrees in cheminformatics or quantum chemistry are preferred. Salaries exceed $130k for candidates with AI integration experience, as highlighted in Computational Chemistry: Mixing Math with Medicine.