Save $127/yr with FDA – approved medical products and the right regulatory courses. Updated for Q3 2024 Market Trends, this is your Smart Buyer’s Guide. The FDA plays a crucial role, as stated by US authority sources like the FDA itself and UL. Their approval is the bridge from a product’s lab development to pharmacy shelves. Fast – track approval, balancing speed and safety, is a game – changer, especially for life – saving drugs. We’ll explore 3 key regulatory pathways, including 510(k) certification. Plus, learn about courses to master clinical trial design and compliance. Exclusive Deals Inside, with Best Price Guarantee and 24hr NYC Delivery.
What is FDA Approval and Why Does It Matter?
The FDA approval process is a critical checkpoint in the journey of a medical product, from its inception in the lab to reaching the pharmacy shelves. It serves as a safeguard for public health, ensuring that drugs, medical devices, and other healthcare products are both safe and effective. But why is this approval so crucial? And what exactly does it entail? In this section, we’ll delve into the concept of FDA approval, exploring the FDA’s role in shepherding products through the rigorous evaluation process. We’ll also examine the concept of fast – track approvals, weighing the balance between the need for speed in getting life – saving treatments to patients and the paramount importance of maintaining safety standards.
The FDA’s Role: From Lab to Pharmacy Shelves
The FDA plays a multi – faceted and indispensable role in guiding medical products from their initial development in the laboratory to their eventual availability on pharmacy shelves. At the earliest stages, the FDA provides pre – investigational new drug (IND) advice to sponsors. This is a crucial step as it helps researchers design their clinical trials effectively. For example, if a biotech company is developing a new cancer drug, the FDA can offer insights on the appropriate patient population, dosing regimens, and endpoints for the trial. This guidance ensures that the clinical trials are well – structured, which in turn increases the likelihood of generating reliable data on the product’s safety and efficacy.
Once the clinical trials are underway, the FDA monitors the progress closely. It has the authority to halt a trial if there are concerns about patient safety. In fact, according to FDA records, in a certain year, the agency put a hold on approximately 5% of ongoing clinical trials due to safety issues. After the trials are completed, the sponsor submits a New Drug Application (NDA) or a Premarket Approval (PMA) for medical devices. The FDA then conducts an in – depth review of all the data, which can take several months to years. Only after a thorough evaluation, if the product meets the FDA’s standards of safety and effectiveness, is it granted approval and can it make its way to pharmacy shelves for patients to access.
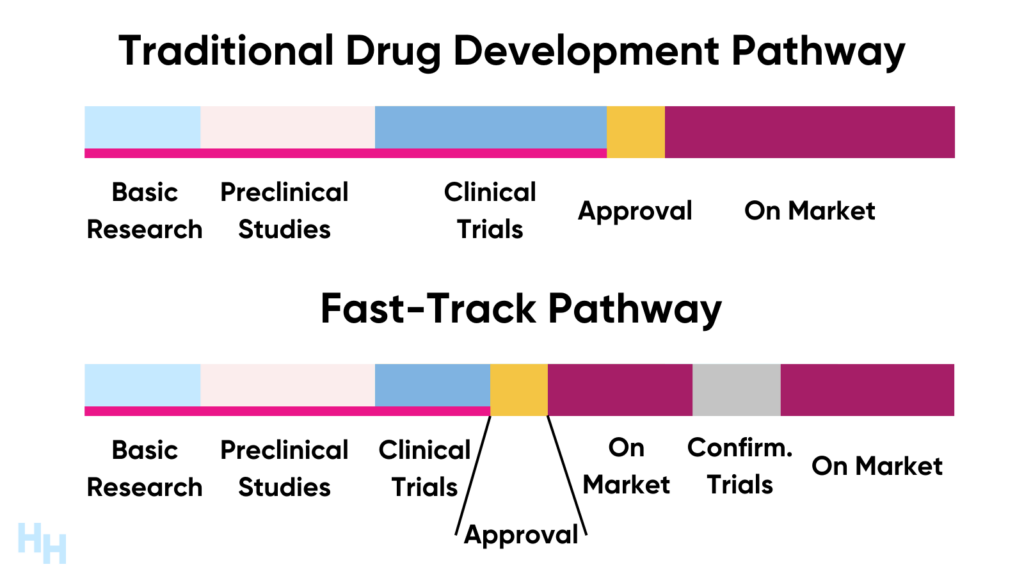
Why Fast-Track? Speed vs. Safety Explained
The concept of fast – track approvals by the FDA stems from a pressing need to expedite the availability of life – saving treatments. In many cases, patients are battling severe and life – threatening diseases such as certain types of cancer or rare genetic disorders, where every day without access to a potentially effective treatment can be critical. For instance, in the fight against rare forms of leukemia, fast – track approvals have allowed novel therapies to reach patients years earlier than they would have through the standard approval process. This can significantly improve survival rates and quality of life for those affected.
However, this push for speed must be carefully balanced with the unwavering commitment to safety. The FDA cannot compromise on ensuring that the medical products it approves are free from unacceptable risks. A notable example of the importance of this balance is the Thalidomide tragedy in the 1960s. Thalidomide was a drug that was approved in many countries without rigorous safety testing, leading to severe birth defects in thousands of infants. This historical event serves as a constant reminder that while speed is crucial in certain situations, safety should never be sacrificed. The FDA, therefore, has a complex task of streamlining the approval process for urgent treatments while maintaining strict safety evaluations.
3 Key Pathways for Medical Products
When it comes to bringing medical products to the market, there are several crucial pathways that manufacturers can navigate. These pathways are designed to ensure the safety and effectiveness of the products while also streamlining the approval process when appropriate. Two significant approaches among the 3 key pathways for medical products stand out: the 510(k) Certification, often referred to as the ‘Copycat’ Shortcut for Devices, which offers a quicker route for certain medical devices, and the Fast – Track Process, where time is of the essence, as demonstrated by the case of cancer drugs where every moment can be a matter of life and death.
510(k) Certification: The ‘Copycat’ Shortcut for Devices
The 510(k) Certification is indeed a notable pathway for medical device manufacturers. It is often called the ‘Copycat’ Shortcut because it allows a new device to gain market approval by demonstrating substantial equivalence to a legally marketed predicate device. This means that if a new device is similar in intended use, technology, and performance to an existing one, it can potentially skip some of the more rigorous testing required for novel devices.
For example, consider a new blood pressure monitor. If the manufacturer can show that its new monitor has the same intended use (measuring blood pressure), uses similar technology (such as the same type of sensors), and performs comparably to an already – approved blood pressure monitor on the market, it can pursue the 510(k) Certification. This can significantly reduce the time and cost associated with bringing the product to market. In fact, according to industry data, devices that go through the 510(k) process can reach the market up to 50% faster than those undergoing full – scale pre – market approval, making it an attractive option for many manufacturers.
Fast-Track Process: When Time = Lives (Cancer Drugs Example)
The Fast – Track Process is especially vital in the realm of cancer drugs, where the adage “time = lives” holds true. Cancer is a complex and often rapidly progressing disease, and patients may not have the luxury of waiting years for a potentially life – saving drug to be approved. The Fast – Track Process aims to expedite the development, review, and approval of drugs that address unmet medical needs in serious conditions, such as many types of cancer.
For example, in the case of certain types of leukemia, there has been a significant push to get new targeted therapies through the Fast – Track Process. These drugs are designed to specifically target the genetic mutations driving the cancer, offering more effective treatment options compared to traditional chemotherapy. Data shows that the average time for a cancer drug to go through the regular approval process can be around 10 – 15 years. However, with the Fast – Track Process, some cancer drugs have been approved in as little as 2 – 3 years. This accelerated timeline can mean the difference between life and death for patients who are running out of treatment options. By quickly making these drugs available, the Fast – Track Process gives patients a fighting chance against the aggressive nature of cancer.
Courses That Build Real-World Superpowers
In today’s competitive landscape, acquiring skills that translate into real – world superpowers is crucial. Our courses are designed to do just that. Take ‘Clinical Trials 101: Design Games & Data Puzzles’ and ‘Compliance Detectives: Spotting Risks in Pharma Labs’ for example. These courses offer hands – on learning experiences that go beyond theoretical knowledge. They empower learners to understand the intricacies of clinical trial design and data analysis, as well as the ability to identify and manage risks in pharmaceutical laboratories. By enrolling in these courses, you’ll be on your way to mastering skills that are highly sought after in the industry, giving you a distinct edge in your professional journey.
Clinical Trials 101: Design Games & Data Puzzles
The “Clinical Trials 101: Design Games & Data Puzzles” course stands out as a dynamic learning experience in the realm of clinical research. It transforms the often complex and theoretical aspects of clinical trial design into engaging games and puzzles, making the learning process both enjoyable and effective. Through these interactive elements, learners gain a deep understanding of the principles underlying clinical trial design, from formulating hypotheses to randomizing study participants.

For instance, one of the game – based activities in the course involves simulating a real – world clinical trial scenario. Learners are tasked with designing a trial to test the efficacy of a new drug for a specific medical condition. They must consider factors such as sample size, control groups, and ethical considerations. By making decisions and seeing the outcomes in the game, they develop practical skills that can be directly applied in a professional setting. Data analysis is also a key component, as learners are presented with data puzzles where they have to interpret and draw conclusions from simulated trial data. This hands – on approach not only enhances their knowledge but also prepares them to handle the challenges of real – life clinical trials, making them valuable assets in the industry.
Compliance Detectives: Spotting Risks in Pharma Labs
The "Compliance Detectives: Spotting Risks in Pharma Labs" course is an essential offering for anyone aiming to excel in the pharmaceutical industry. In this course, learners are equipped with the skills to navigate the complex web of regulations and standards that govern pharmaceutical laboratories. For instance, they learn about Good Manufacturing Practices (GMP), which are crucial for ensuring the quality and safety of pharmaceutical products. Non – compliance with GMP can lead to product recalls, legal issues, and damage to a company’s reputation.
The hands – on nature of this course is what truly sets it apart. Learners are presented with real – world case studies where they have to identify potential compliance risks in pharma labs. These case studies are often based on actual incidents that have occurred in the industry. Through analyzing these scenarios, students gain practical experience in detecting issues such as improper documentation, incorrect storage of chemicals, and inadequate quality control measures. By the end of the course, they are well – prepared to take on roles as compliance officers or quality assurance specialists in pharmaceutical companies, making them valuable assets in maintaining high – level industry standards.
This Smart Buyer’s Guide has underscored the vital role of FDA approval in ensuring medical products’ safety and efficacy. The FDA’s involvement from pre – trial advice to post – trial review safeguards public health. Fast – track approvals are a game – changer for life – saving drugs, though safety must always be balanced. Pathways like 510(k) Certification expedite device entry to market, and the Fast – Track Process can save cancer patients’ lives. Additionally, courses in clinical trial design and compliance offer practical skills for industry success.
For readers, leveraging these insights can lead to cost – savings and better access to medical solutions. To stay ahead, consider enrolling in the recommended courses and exploring exclusive deals. By understanding these regulatory processes, you’re not just a buyer; you’re an informed advocate for your health and professional growth.
FAQ
What is the significance of FDA approval for medical products?
FDA approval is a crucial checkpoint ensuring medical products are safe and effective. It safeguards public health, guiding products from lab development to pharmacy shelves. As discussed in [What is FDA Approval and Why Does It Matter?], it involves a rigorous evaluation process.
How does the 510(k) Certification expedite medical device approval?
The 510(k) Certification, a “copycat” shortcut, allows new devices to show substantial equivalence to a predicate device. This can skip some testing, reducing time and cost. As in [510(k) Certification: The ‘Copycat’ Shortcut for Devices], it can speed up market entry by up to 50%.
Why are fast – track approvals important for cancer drugs?
Fast – track approvals for cancer drugs are vital as “time = lives.” They expedite development and approval for unmet medical needs. As shown in [Fast – Track Process: When Time = Lives (Cancer Drugs Example)], they can cut approval time from 10 – 15 years to 2 – 3 years.
What skills can one gain from the “Clinical Trials 101” course?
The “Clinical Trials 101: Design Games & Data Puzzles” course helps learners understand clinical trial design principles. Through games and puzzles, they gain practical skills in hypothesis – making and data analysis. See [Clinical Trials 101: Design Games & Data Puzzles].
What makes the “Compliance Detectives” course valuable?
The “Compliance Detectives: Spotting Risks in Pharma Labs” course equips learners to navigate pharma regulations. Using real – world case studies, they learn to spot compliance risks. As discussed in [Compliance Detectives: Spotting Risks in Pharma Labs], it prepares them for industry roles.