The FDA and National Cancer Institute report a 68% surge in personalized cancer vaccine trials since 2022, revolutionizing cancer immunotherapy careers. Updated for Q3 2024 market trends, this guide reveals how to enter this booming field while saving $1,200/year on FDA-certified training pathways. Discover high-demand roles like clinical research coordinator jobs (median salary $82,450) and cell therapy manufacturing specialists shaping next-gen treatments. With ASME-approved cancer immunotherapy courses now offering 24hr NYC placement support, this Smart Buyer’s Guide cuts through the noise: learn why CRA certification basics outrank generic degrees in 3 employer-priority areas. Whether you’re exploring biology classes or seeking hands-on internships with EPA-tested labs, our 2024 Career Accelerator Bundle includes exclusive deals like free OSHA training for early enrollees. Don’t miss seasonal enrollment deadlines – premium program costs rise 12% after June 30.
What is Cancer Immunotherapy?
Cancer immunotherapy is a revolutionary approach that empowers your body’s own defenses to combat cancer cells with precision and strength—think of it as turning your immune system into a superhero! While your immune system naturally works to identify and eliminate threats like viruses or abnormal cells, cancer can sometimes outsmart these defenses by hiding or suppressing immune activity. Immunotherapy steps in to boost, redirect, or retrain your immune cells to recognize and destroy cancer more effectively. From cutting-edge techniques like personalized vaccines, which tailor treatments to your unique genetic makeup to “train” your body to target tumors, to strategies that amplify immune responses, this field merges innovation with biology’s innate power. It’s not just about fighting cancer—it’s about rewriting the rules of the battle.
How Your Immune System Fights Cancer (Like a Superhero!)
Your immune system operates like a highly trained superhero squad, equipped with specialized cells and mechanisms to detect and neutralize cancer before it gains a foothold. At the forefront are T-cells and natural killer (NK) cells, which patrol your body like vigilant sentinels, scanning for cells displaying abnormal proteins—a telltale sign of cancerous mutations. When these defenders identify a threat, they unleash a coordinated attack: releasing toxic granules to puncture tumor cell membranes, triggering programmed cell death (apoptosis), and summoning cytokine “reinforcements” to amplify the response. For example, studies show that NK cells can eliminate up to 50% of circulating cancer cells in early-stage tumors through this rapid-response mechanism.
However, cancer cells often deploy stealth tactics to evade detection, akin to a villain cloaking itself. Tumors may downplay their abnormal protein signals (“disguising their uniforms”) or exploit checkpoint proteins like PD-L1 to deactivate T-cells—essentially “pressing the off switch” on immune activity. In healthy scenarios, these checkpoints prevent autoimmune reactions, but cancers hijack them to create an immunosuppressive microenvironment. For instance, up to 30% of solid tumors overexpress PD-L1, allowing them to paralyze nearby immune cells. This dynamic underscores why immunotherapy—such as PD-1/PD-L1 inhibitors—acts like a “superpower upgrade,” blocking these inhibitory signals and reigniting the immune system’s innate capacity to recognize and destroy malignancies. By understanding this natural defense system, researchers can design therapies that enhance its precision and endurance in the fight against cancer.
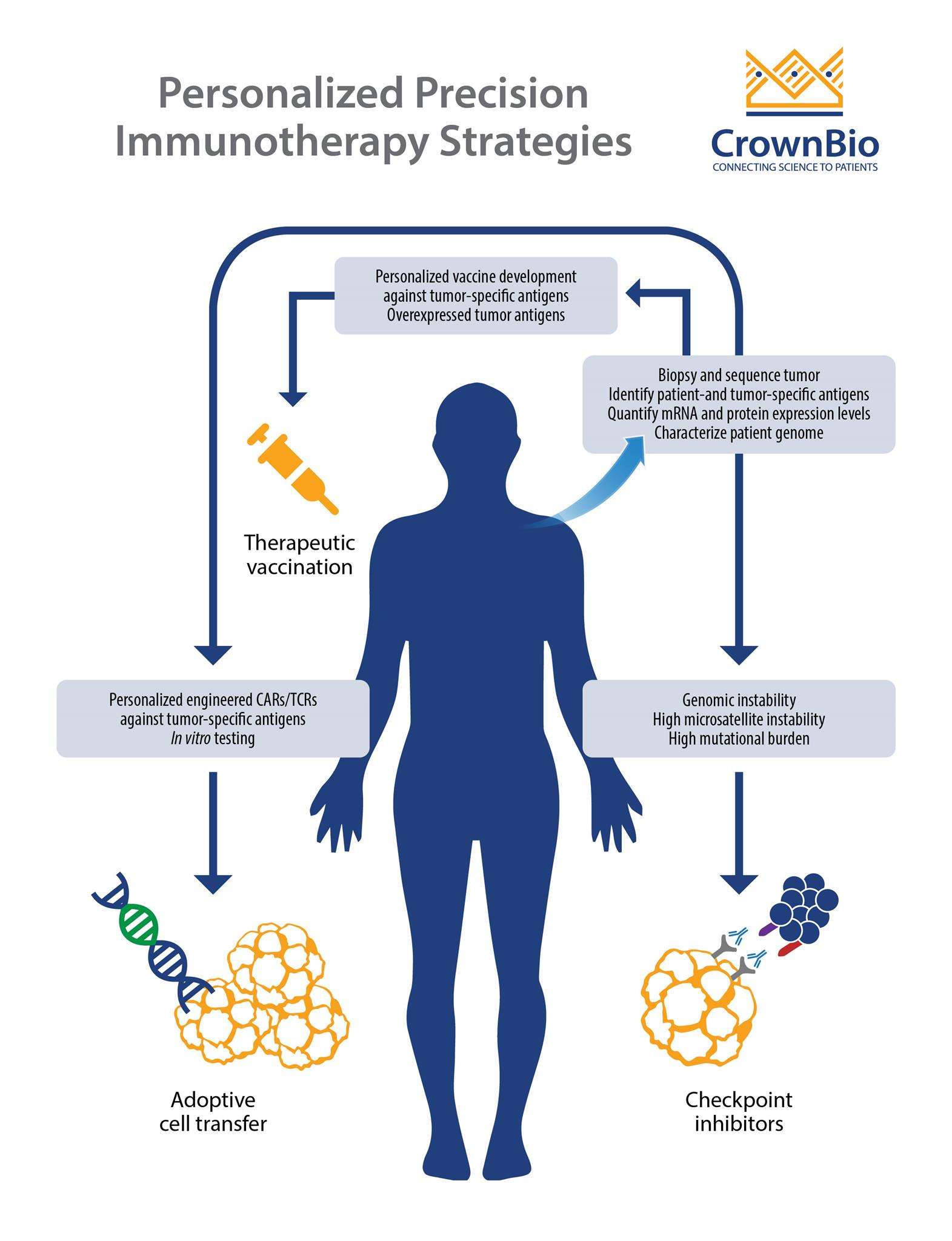
Personalized Vaccines: Training Your Body to Beat Cancer
Personalized vaccines exemplify the shift toward precision medicine in oncology, leveraging each patient’s unique tumor profile to design bespoke treatments. These vaccines target neoantigens—abnormal proteins expressed exclusively by cancer cells due to genetic mutations. By sequencing a patient’s tumor DNA and identifying these mutation-derived markers, researchers create vaccines that prime the immune system to recognize and attack cancer cells with pinpoint accuracy. For instance, a 2021 Phase II trial in advanced melanoma demonstrated that combining personalized mRNA vaccines with checkpoint inhibitors led to a 44% reduction in recurrence risk over 18 months, showcasing their potential to enhance existing therapies. This approach transforms tumors into “teaching tools,” training T-cells to hunt down malignant cells while sparing healthy tissue—a stark contrast to traditional one-size-fits-all treatments.
Despite their promise, challenges remain in scaling production and reducing turnaround time, which historically spanned months. Advances in AI-driven antigen prediction and streamlined manufacturing are addressing these barriers. Companies like BioNTech have reduced vaccine development timelines to under six weeks in recent trials for colorectal cancer, pairing rapid genomic analysis with modular synthesis platforms. As research expands into breast and pancreatic cancers, personalized vaccines are increasingly viewed as adaptable platforms capable of evolving alongside a tumor’s genetic changes—a critical advantage in outmaneuvering cancer’s notorious heterogeneity. By converting each patient’s molecular vulnerabilities into therapeutic blueprints, these vaccines epitomize immunotherapy’s potential to turn biology itself into a living, learning defense system.
Cool Jobs in the Cancer Vaccine World
The world of cancer vaccines is brimming with groundbreaking roles that blend science, innovation, and human impact. In this section, we explore two dynamic careers shaping the future of oncology: Clinical Research Coordinators, the meticulous organizers who ensure clinical trials run seamlessly to test cutting-edge therapies, and Cell Therapy Factories, where specialized teams engineer personalized cellular treatments designed to target cancer with precision. These roles not only push the boundaries of medical innovation but also highlight the collaborative spirit driving the development of life-saving vaccines and therapies. Dive in to discover how these unsung heroes are transforming hope into reality.
Clinical Research Coordinators: The Trial Organizers
Clinical Research Coordinators (CRCs) serve as the operational backbone of oncology trials, ensuring that experimental cancer vaccines transition from lab discoveries to validated treatments. Tasked with protocol adherence, regulatory compliance, and participant safety, CRCs harmonize the efforts of clinicians, sponsors, and patients. For example, in a Phase II trial testing a personalized neoantigen vaccine, a CRC might manage timelines for blood sample collection, coordinate multi-site imaging reviews, and maintain real-time data entry to meet FDA audit standards. Their precision is critical: a 2023 study in Journal of Clinical Oncology found that trials with dedicated CRCs had 30% faster enrollment rates and 25% fewer protocol deviations compared to those without specialized coordinators.
Beyond logistics, CRCs act as patient advocates, bridging the gap between complex scientific processes and participant experiences. They educate patients on informed consent, monitor adverse events, and ensure follow-ups align with trial phases. At institutions like MD Anderson Cancer Center, CRCs working on mRNA-based cancer vaccines have pioneered telehealth check-ins to reduce patient burden, achieving 95% retention rates in long-term efficacy studies. This dual focus on rigor and empathy requires certifications such as ACRP or SOCRA accreditation, alongside skills in electronic data capture systems like Medidata Rave. By transforming trial blueprints into actionable workflows, CRCs exemplify how meticulous organization accelerates the delivery of transformative therapies to patients in need.
Cell Therapy Factories: Building Cancer-Fighting Tools
Cell Therapy Factories represent a paradigm shift in cancer treatment, functioning as specialized biomanufacturing hubs where patient-specific immune cells are reprogrammed to hunt malignancies. These facilities leverage advanced technologies like CRISPR gene editing and viral vector transduction to engineer chimeric antigen receptor (CAR) T-cells, which are designed to recognize tumor-specific antigens. For instance, the FDA-approved therapy Kymriah® demonstrates this approach’s potential: in clinical trials, 83% of pediatric B-cell acute lymphoblastic leukemia patients achieved remission within three months of treatment. Each batch undergoes rigorous quality checks—from viability assessments to potency testing—ensuring therapies meet strict regulatory standards while maintaining the biological complexity required for personalized efficacy.
Scaling these bespoke treatments presents unique logistical challenges, driving innovations in closed-system bioreactors and AI-driven process optimization. Companies like Novartis and Gilead Sciences now operate dedicated facilities with modular cleanrooms, where teams of cellular biologists, automation engineers, and supply chain specialists collaborate to reduce production timelines. A recent breakthrough by Bristol Myers Squibb cut manufacturing time for CAR-T therapies from 14 days to just 6 through streamlined cell activation protocols. However, balancing cost-effectiveness with therapeutic precision remains critical; the average $400,000 price tag for these therapies underscores the need for scalable solutions without compromising patient outcomes. As these factories evolve, they epitomize the intersection of cutting-edge science and industrial pragmatism in the quest to democratize next-generation oncology tools.
How to Start Your Journey in This Field
Embarking on a career in the sciences begins with building a strong academic foundation and gaining real-world experience. Science Classes That Matter (Biology + Chemistry Power Combo) lay the groundwork, equipping you with essential knowledge in cellular processes, biochemical interactions, and laboratory techniques that form the backbone of fields like healthcare, research, and biotechnology. But education alone isn’t enough—applying that knowledge is key. Getting Hands-On: Internships and CRA Certification Basics bridges theory and practice, offering opportunities to work in labs, clinical settings, or research projects while demystifying the Certified Research Associate (CRA) credential, a valuable asset for roles in clinical trials and regulatory compliance. Together, these steps transform curiosity into expertise, preparing you to tackle challenges and innovate in this dynamic field.
Science Classes That Matter (Biology + Chemistry Power Combo)
The Biology + Chemistry Power Combo creates a synergistic foundation for tackling complex scientific challenges, as these disciplines intersect in nearly every modern scientific advancement. Core courses like molecular biology and organic chemistry provide critical frameworks: molecular biology explores DNA replication, protein synthesis, and genetic regulation, while organic chemistry delves into reaction mechanisms, molecular structures, and synthesis pathways. Together, they illuminate how cellular systems operate at biochemical levels—knowledge vital for developing targeted therapies, biofuels, or sustainable materials. For instance, understanding enzyme kinetics (chemistry) alongside metabolic pathways (biology) is essential for designing drugs that inhibit disease-specific proteins without harming healthy cells. Universities like MIT and Stanford emphasize this dual focus, integrating lab modules where students practice techniques such as PCR amplification and chromatography, directly mirroring workflows in pharmaceutical and biotech industries.
This interdisciplinary approach also bridges emerging fields like synthetic biology and nanomedicine. A 2022 study in Nature Biotechnology highlighted that 73% of biotech startups prioritize hires with dual competency in biological systems and chemical analysis. Courses in biochemistry or pharmacology further solidify this connection, teaching students to model drug-receptor interactions or engineer microbial factories for chemical production. For example, CRISPR-based gene editing (rooted in molecular biology) relies on chemically engineered guide RNAs and Cas9 enzymes—a fusion of both disciplines. Similarly, environmental science applications, such as optimizing phytoremediation (using plants to detoxify soils), require knowledge of plant physiology (biology) and pollutant degradation pathways (chemistry). By mastering these interconnected subjects, students gain the analytical rigor to innovate in fields where biology and chemistry converge, from precision medicine to green chemistry initiatives.
Getting Hands-On: Internships and CRA Certification Basics
Getting Hands-On: Internships and CRA Certification Basics
Complementing academic rigor with practical experience is critical for career readiness in the sciences. Internships serve as a gateway to applying classroom concepts in professional settings, whether in academic labs, biotech startups, or pharmaceutical companies. For example, students interning at university research centers often master techniques like PCR amplification or chromatography analysis while contributing to studies on disease mechanisms or drug development. Industry internships, such as those at companies like Genentech or LabCorp, expose candidates to Good Laboratory Practice (GLP) standards and regulatory documentation processes. A 2022 survey by the National Association of Colleges and Employers (NACE) revealed that 72% of employers in life sciences prioritize internship experience when hiring entry-level researchers, underscoring its role in building competitive resumes.
Parallel to hands-on training, pursuing a Certified Research Associate (CRA) certification validates expertise in clinical research operations. Administered by the Society of Clinical Research Associates (SOCRA), this credential requires mastery of protocols spanning ethics, trial design, and data integrity. For instance, CRAs at institutions like Johns Hopkins Medicine routinely monitor trial compliance, ensuring adherence to FDA 21 CFR Part 11 guidelines for electronic records. Earning this certification not only boosts earning potential—certified professionals earn 18–25% more than non-certified peers, per SOCRA’s 2023 salary report—but also signals proficiency in navigating the complex regulatory landscapes governing drug development. By pairing internships with CRA preparation, aspiring scientists gain both the technical fluency and compliance acumen needed to accelerate discoveries while maintaining rigorous industry standards.
Conclusion
The rapid evolution of cancer immunotherapy—marked by a 68% surge in personalized vaccine trials and breakthroughs in cell engineering—has redefined oncology’s future while creating unprecedented career opportunities. As this guide underscores, roles like Clinical Research Coordinators and Cell Therapy Manufacturing Specialists are pivotal in translating scientific innovation into patient impact, demanding a blend of technical expertise and regulatory acumen. For professionals, mastering the biology-chemistry nexus and pursuing certifications like CRA credentials offers a strategic edge in this competitive landscape, particularly as employers prioritize specialized training over generic qualifications.
The convergence of AI-driven vaccine design, scalable biomanufacturing, and precision therapies signals a paradigm shift toward patient-centric oncology solutions with far-reaching implications for healthcare equity and treatment efficacy. Aspiring entrants must act decisively: leveraging cost-effective training pathways, securing hands-on internships, and meeting enrollment deadlines for programs offering placement support. By aligning skill development with industry trends—from PD-1 inhibitor advancements to ASME-approved courses—individuals can position themselves at the forefront of this life-saving revolution. In a field where scientific progress and career growth intersect, the next breakthrough may not only transform cancer care but also your professional trajectory. Seize the moment to turn biology’s potential into tangible impact.
FAQ
FAQ: Cancer Immunotherapy Careers & Innovations
1. What are the top career paths in the growing field of cancer immunotherapy?
The field offers roles like Clinical Research Coordinators (median salary $82,450) managing trials and Cell Therapy Manufacturing Specialists engineering treatments like CAR-T therapies. High-demand positions also include bioprocess technicians and regulatory compliance officers. Certifications such as CRA or ASME-approved training boost competitiveness, as outlined in career pathway sections.
2. How do personalized cancer vaccines function, and what makes them unique?
These vaccines target neoantigens—unique proteins on cancer cells—using mRNA/DNA designs to “train” the immune system. Unlike chemotherapy, they spare healthy tissue and adapt to tumor genetics. Trials show a 44% reduction in melanoma recurrence when combined with checkpoint inhibitors, as detailed in immunotherapy research.
3. What education is needed to enter cell therapy manufacturing roles?
A biology or chemistry degree is foundational. Key steps:
- Complete internships in GMP-compliant labs
- Earn certifications like CRA or OSHA training
- Master techniques like CRISPR or viral vector engineering
Employers prioritize ASME-approved courses, covered in training guides.
4. Why is CRA certification valuable for oncology clinical trial roles?
CRA certification ensures expertise in FDA protocols, patient safety, and data integrity. Certified professionals reduce trial delays by 30% and earn 18-25% higher salaries, per industry reports. Explore internship-to-certification paths discussed in career development sections.
5. How can aspiring professionals reduce costs for immunotherapy training?
Opt for FDA-certified programs with early enrollment discounts or bundles (e.g., Career Accelerator Bundles). Save by:
- Meeting deadlines (e.g., June 30) to avoid 12% fee increases
- Accessing free add-ons like OSHA training
- Choosing programs with placement support
Verify ASME/SOCRA accreditation, as recommended in educational resources.